The international multicenter frontline therapy trial AIEOP-BFM ALL 2017 prospectively randomizes CD19-targeting T-cell engager blinatumomab (BLN) against standard of care (SOC) to improve outcomes in pediatric patients (pts) with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) stratified for high (HR) and medium (MR) risks of relapse. In HR arm BLN is given earlier, directly after intensified HR therapy block, while in the MR arm, it is given towards the end of chemotherapy, before maintenance. The immune composition prior to BLN might thus differ between both treatment groups (HR vs MR) which may impact BLN-mediated anti-leukemic activity.
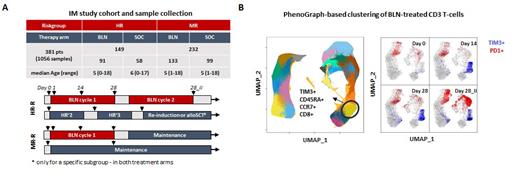
Within the Germany-wide immune monitoring program accompanying the AIEOP-BFM ALL 2017 trial we addressed this question and studied a total of 1056 longitudinal peripheral blood samples collected between 11/2018 and 05/2022 from 381 BCP-ALL pts. Samples were prospectively analyzed with multicolor flow cytometry to study immune cell composition (Panel 1) at the time of BLN application (d0) and under ongoing immunotherapy within first 24h (d1), at day 14 (d14), at the end of the cycle I (d28) and end of cycle 2 (day28_II), if applicable (Figure 1A). In-depth T-cell profiling was performed in the BLN arm, utilizing 4 additional 12-color panels concentrated on markers associated with T-cell functionality (measured with standardized cytometer settings). Data were analyzed using classical 2D-manual gating strategies (> 2000 variables) and completed with R-based machine learning algorithms; namely partial Least-Squares Discriminant Analysis (PLS-DA), a multivariate dimensionality-reduction tool for identifying HR- and MR-defining immune signatures and PhenoGraph algorithm for unsupervised T-cell cluster detection. T-cell receptor excision circles (TRECs) and TR-repertoire analysis were also performed among paired samples of 117 BLN-treated pts.
Baseline immune composition differed notably between HR and MR pts directly prior to BLN therapy, as shown by PLS-DA analysis on manually gated immune subsets. HR pts showed higher neutrophil counts and lower lymphocyte and monocyte counts compared to MR pts. Interestingly, MR pts had higher levels of B- and T-cells, which are crucial for BLN-mediated T-cell activation. HR pts displayed an elevated CD4+/CD8+ ratio, shifted naïve/memory T-cell phenotypes and increased proportions of immunosuppressive T-regulatory cells. These results suggest that more intensive therapy protocols directly prior to BLN influence the immune composition towards compromised states in the HR arm, potentially affecting BLN-mediated T-cell leukemia control. BLN effectively depleted CD19+ cells in 221 out of 224 pts. We observed significantly stronger T-cell activation/expansion in MR pts compared to HR pts under BLN therapy. This expansion that was primarily attributed to naïve CD8+ T-cell populations, resulting from clonal T-cell proliferation independent of thymic contribution, as supported by TREC and TR-repertoire analysis. The PhenoGraph clustering also identified this expanding naïve CD8+ T-cell populations, surprisingly marked by loss of adhesion molecule DNAM1 (panel 1), normally involved in NK and T-cell mediated cytotoxicity and by gaining the expression of TIM-3 (panel 3), an inhibitory molecule usually co-expressed with PD1 on exhausted T-cells with memory phenotypes. In this setting however, TIM3 was not co-expressed with PD1 (Figure 1B) and its expression was correlated with the B-cell levels at baseline and was strongest in MR pts under BLN therapy. Our data suggest an until now undescribed association between DNAM1 and TIM3 with BLN-mediated T-cell activation, hinting that they may serve as a measure of T-cell activation strength or an early T-cell exhaustion marker.
In conclusion our study provides clinically relevant insights in how chemotherapy prior BLN might affect the patients´ immune system, possibly influencing the BLN anti-leukemic activity, and helps to determine the optimal place of immunotherapy with BLN in multimodal treatment schedules. Here, we report until know undescribed effect of BLN on T-cell compartment. Upcoming clinical data, which will be correlated with obtained immune profiles will answer the open question regarding the clinical relevance of these findings, and shed light into the mechanisms determining response to BLN, for its maximum benefit in childhood ALL.
Disclosures
Bendig:Amgen: Research Funding. Bastian:BeiGene: Other: Travel Honoraria. Ritgen:Janssen: Consultancy, Honoraria; Roche: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy, Honoraria, Other: travel support; Abbvie: Consultancy, Research Funding. Baldus:Amgen: Consultancy; AstraZeneca: Consultancy; BMS: Consultancy; Astellas: Consultancy; Jazz Pharmaceuticals: Consultancy; Gilead: Consultancy; Jannsen: Consultancy. Cario:Servier, Amgen: Research Funding; JazzPharma: Speakers Bureau. Rossig:Amgen, BMS, Novartis, Pfizer, Roche, MSD: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal